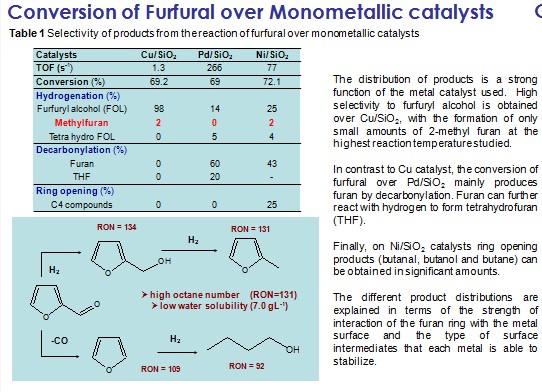
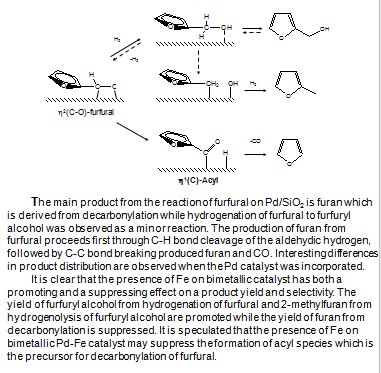
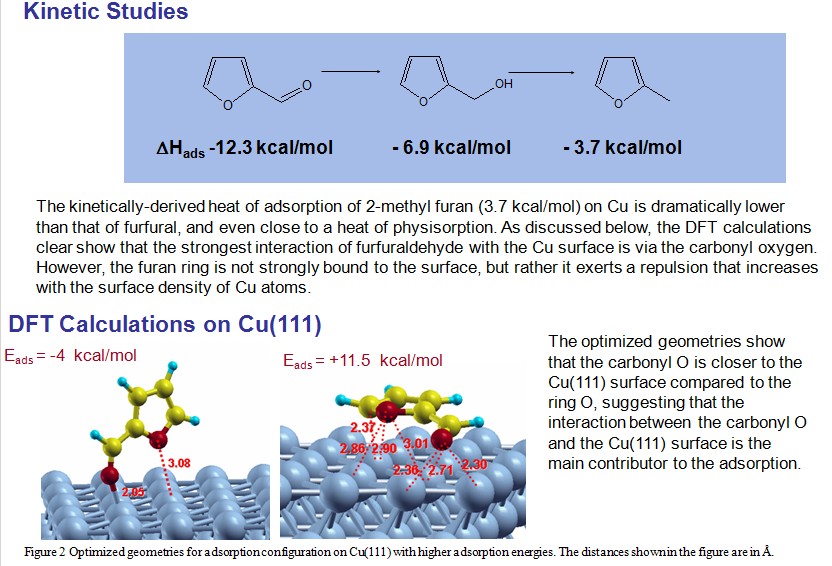
Molecular engineering of biofuel components requires a fine control of the surface chemistry to maximize the yield of the desired molecules. This study illustrates how a detailed knowledge of the nature of adsorbed species on different metal surfaces leads to tailored catalysts with the desired properties. In this case deoxygenation of biofuel components has been achieved with minimal reduction of carbon yield. For example, monometallic Pd catalyst is highly active for decarbonylation of furfural, a main aromatic aldehyde of biomass-derived feedstock, by C-C bond cleavage yielding furan and CO as the dominant product. However, the role of the bio-oil upgrading process is not only to eliminate O, but also to retain C in the liquid preferably by the cleavage of C-O bond. In this regard, bimetallic alloy catalysts may be interesting systems because the typically strong interaction of oxygen atom with Group VII metals can be increased. For example, the addition of Fe to Pd forming Pd-Fe alloy can dramatically change the product selectivity from furan to methyl furan. This can be explained by a stronger interaction of the Pd-Fe bimetallic catalysts with the O atom, making the C-O bond cleavage more favorable, leading to production of methylfuran.