MgO-containing catalysts are known to be active in aqueous-phase aldol condensations of ketones and furanic aldehydes, which are reactions with a high potential in a future bio-oil upgrading process scheme. The model reaction of acetone and furfural has been studied in several papers, but the effects of acid, alcohol and other organics, which are abundant in bio-oil, on the catalysis and the catalyst are not clear. In order to evaluate the potential of MgO-based catalysts for bio-oil upgrading, acetic acid, methanol, and tetrahydrofuran (THF) were introduced into the reaction mixture to simulate a fraction of bio-oil, and the effect of these components on the aldol condensation of furfural and acetone was analyzed. The reusability of these catalysts was also studied.
ATR-IR spectra of the supernatant after oxides were treated with acetic acid solution at 80°C for 24 h:

The activities of MgO-based catalysts decrease with acid concentration, and the catalysts lost most of their activity when the concentration of acetic acid exceeded 3 wt.%
Effect of THF (250 ppm BHT as inhibitor) as solvent component on the conversion of furfural with different catalysts. T=80C:

Effect of CH3OH as solvent component on the conversion of furfural with different catalysts. T=80 °C:

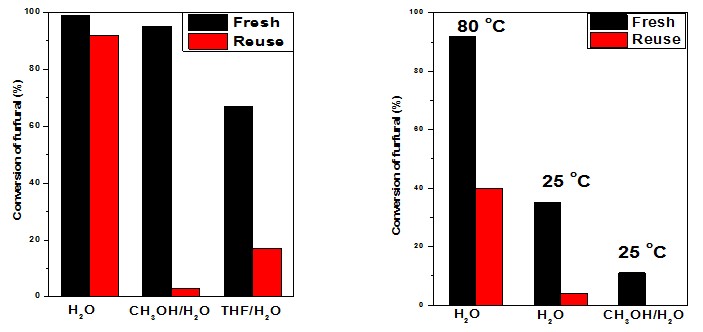
Reutilization of Mg-AlOx (850C) (left) and MgO (right) in aldol condensation. After the 1st run, the catalysts were recovered by centrifuging, washing with acetone and H2O, and drying at 100C for 1 h.