
Lefranc, Agnès, and J. Bundgaard. 2000. Controlled variation of body size by larval crowding in Drosophila melanogaster. Dros. Inf. Serv. 83:171-174.
|
|
Lefranc, Agnès, and J. Bundgaard. 2000. Controlled variation of body size by larval crowding in Drosophila melanogaster. Dros. Inf. Serv. 83:171-174. |
|||
Controlled variation of body size by larval crowding in Drosophila melanogaster.
Lefranc, Agnès, and J. Bundgaard. University of Aarhus, Department of Ecology and Genetics, Building 540, Ny Munkegade, DK-8000 Aarhus-C, Denmark. Correspondence: Agnès Lefranc, Laboratoire Fonctionnement et Evolution des Systèmes Ecologiques, UMR 7625, 7 Quai St Bernard, Case 237, 75252 Paris Cédex 05. FAX: 33 1 4427 3516; E-mail: Agnes.Lefranc@snv.jussieu.fr.
Abstract
Body size variation as a function of nutrition during larval stages
was studied in an inbred strain of Drosophila melanogaster. We confirmed that body size varies with
larval crowding, but we also found significant size variation with developmental
time at all crowding levels. Based on these experiments, we present a method whereby one
can obtain distinct size classes for experimental purposes.
Introduction
Starvation during larval development is known to be one of several
environmental factors influencing body size in Drosophila melanogaster (Robertson, 1960; Miller, 1964).
It has been discussed whether variation in body size following larval
starvation is due to selection or phenotypic plasticity in response to environmental
conditions (Robertson, 1960). The purpose of the present experiments was to create a number
of (non-overlapping) size classes that could be used for further experiments
studying possible fitness effects of body size in D. melanogaster. We therefore used an inbred laboratory strain, thereby minimizing
genetic variation for body size on which larval selection could act.
Material and Methods
Flies used in the experiments were from an Oregon-R wild type strain originally procured from California Institute of Technology, California, USA. Prior to the experiments the stock had been maintained for many years in vials/bottles and were therefore highly inbred with little genetic variation. The experimental medium was a yeast-agar-sugar medium: 5 litres of medium contain 160 grams dried yeast, 70 grams agar, 270 grams sugar, water and three teaspoons of methyl-4-hydroxybenzoate. All experiments were carried out at 25oC, under constant light and ca. 50% relative humidity.
One to two thousand newly-emerged flies were placed in a plexiglas
box containing petri dishes with yeasted food.
The flies were aged for 2-3 days.
Petri dishes with a layer of medium, lightly sprinkled with dry yeast,
were then introduced into the cages for oviposition for 5 hours. The first instar larvae emerged approximately
22 hours later and were then collected on needles and gently transferred to
vials containing different amounts of medium, representing four different
crowding levels (i.e., 400 larvae on
2, 3, 4 or 5 ml of medium per vial, respectively), and an uncrowded control
with 30 larvae per vial on 7 ml of medium with extra yeast added. In each vial the medium was layered on
top of a complementary 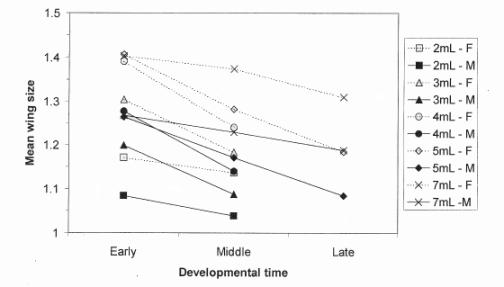
Figure 1. Mean wing size for each sex (males M and
females F), each starvation level and each developmental time.
volume
of 1.5% agar solution in order to have a total
Newly-emerged flies were collected daily. The flies were counted and stored in vials with fresh medium until their wing measure was taken. Counting imagos allowed us to calculate survival rates for each starvation level. The total emergence period was arbitrarily divided up into three intervals: early (from the beginning of the hatching period to 300 h after the transfer of the larvae), middle (from 300 h to 375 h after the transfer of the larvae), and late (from 375 h after the transfer of the larvae until the end of the hatching period).
For each starvation treatment, each replicate and each interval, a maximum of 30 males and females were randomly selected and used for the analyses. The flies were killed by etherization and their wings were pulled off and placed in a drop of water on a microscope slide covered with a cover slip. Wing length was measured from the intersection between the anterior cross vein and the third longitudinal vein (L3) to the intersection between L3 and the distal wing margin using a binocular microscope with a digital filar eyepiece (LASICO: Los Angeles Scientific Instrument Company, Inc., USA). For flies emerging during the middle period, thorax length was also measured, from the posterior tip of the scutellum to the anterior edge of the pronotum.
Results and Conclusions
Larva-to-adult survival was decreasing with the degree of starvation: from 84% for flies on 7 ml of medium (uncrowded) to 50% on 5 ml, 17% on 4 ml, 11% on 3 ml and 4% for larvae on 2 ml of medium.
Wing size variation between replicates for each starvation level and each developmental time was not significant (one way ANOVA on replicate effect, within each crowding and developmental time levels, all p > 0.05). We found a strong correlation between wing and thorax length for flies that emerged during the middle period (r = 0.428, n = 30, P < 0.05) in accordance with Robertson and Reeve (1952).
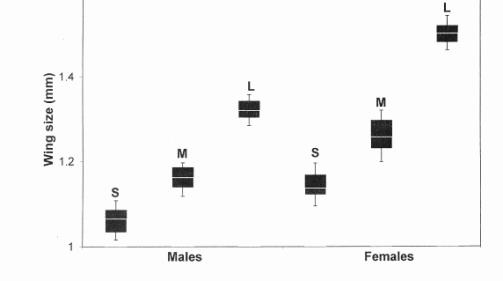 |
| Figure 2. Distribution of wing sizes of small (S), medium (M) and large (L). The box includes 50%, and the vertical bars 90% of the variation. The white horizontal line through the box corresponds to the value of the median. |
Because of the considerable size variation with developmental time for both sexes at all crowding levels, we decided only to use flies emerging during the middle of the hatching period. In this way size variation within each starvation level could be kept at a minimum, and overlap between different size classes was prevented. It is hence possible to create three distinct size classes by applying the following protocol (see Figure 2):
· Large (L) flies were obtained by transferring 30 first instar larvae to a vial containing 7 ml of medium sprinkled with dry yeast. These conditions can be considered as non-crowded (control) conditions.
· Medium (M) flies were obtained by transferring 400 first instar larvae to a vial containing 5 ml of medium (layered on top of 2 ml of agar solution).
· Small (S) flies were obtained by transferring 400 first instar larvae to a vial containing 3 ml of medium (layered on top of 4 ml of agar solution).
The flies obtained with this method can then be used for further studies, i.e. of the effect of size variation on various life-cycle components, including behavioural ones, without artifacts due to hidden genetic variation.
References: Miller, R.S., 1964, Ecology, 45:132-148; Robertson, F.W., 1960, Genet. Res. Camb. 1:305-318; Robertson, F.W., and E.C.R. Reeve 1952, J. Genet. 50:414-448.