
Buratovich, Michael A., and Elizabeth L. Wilder. 2001. The role of wingless in the development of the proximal wing hinge of Drosophila. Dros. Inf. Serv. 84: 145-157.
|
|
|
|||
|
|
||||
The role of wingless in the development of the proximal wing hinge of Drosophila.
Buratovich, Michael A.1, and Elizabeth L. Wilder.2 1Department of Biochemistry, Spring Arbor University, Spring Arbor, MI 49283; 2Department of Cell and Developmental Biology, University of Pennsylvania School of Medicine, Philadelphia, PA 19104; phone 517-750-6383; fax 517-750-2108; michaelb@arbor.edu
Abstract
We have examined
the role of Wingless (Wg) in the development of the proximal portion of the
Drosophila wing hinge. We find that in the dorsal hinge, ectopic
expression of wg causes duplication
of Axillary Sclerite 3 (AS3) at the expense of Axillary Sclerite 1 (AS1),
Axillary Sclerite 2 (AS2) and the Unnamed Plate (UP). Attenuation or removal
of Wg activity causes shrinkage or deletion of AS3. Therefore Wg is necessary
to establish the development of AS3 in the dorsal hinge. In the ventral hinge,
ectopic expression of Wg removes the Axillary Pouch (AP) and attenuation or
removal of Wg activity in the ventral hinge duplicates the AP. Therefore,
Wg is necessary to restrict the formation of the AP to its particular location
in the ventral hinge. Thus Wg is an important signaling molecule in the development
of the proximal dorsal and ventral wing hinge.
Introduction
Wnt proteins constitute a large family of secreted glycoproteins that function as signaling molecules during the development of both invertebrate and vertebrate organisms (for review see Cadigan and Nusse, 1997) . Wnt genes show very widespread expression and many tissues require Wnt proteins for their development.
Wg signaling studies in Drosophila have made great contributions to our understanding of Wnt signal transduction. Wnt proteins associate with the Frizzled (Fz) family of receptors (Bhanot et al., 1996) . The Wnt signal is then transduced by a cytoplasmic protein, Dishevelled (Dsh; Klingensmith et al., 1994; Theisen et al., 1994) , resulting in repression of the Zeste-white3/Shaggy kinase (Siegfried et al., 1990; Siegfried et al., 1992) , the ortholog of Glycogen Synthase Kinase 3b. Repression of the Shaggy kinase involves the dissociation of a complex between Shaggy and a scaffold protein called Axin (Hamada et al., 1999; Ruel et al., 1999; Willert et al., 1999) , which also acts as a negative regulator of Wg signaling. The Shaggy/Axin complex binds to the Armadillo protein, which is the ortholog of b-catenin (Peifer and Wieschaus, 1990) , and phosphorylates it (Peifer et al., 1994a; Peifer et al., 1994b) , which marks Armadillo for degradation (Pai et al., 1997) and holds it in within the cytoplasm (Tolwinski and Wieschaus, 2001) . Inactivation of Shaggy stabilizes Armadillo protein (Peifer et al., 1991) , which translocates to the nucleus where it associates with Pangolin, the Drosophila LEF-1/TCF ortholog (Brunner et al., 1997; van de Wetering et al., 1997) to regulate gene expression. Vertebrate orthologs of these proteins have been shown to function in a similar cascade to transduce Wnt-1-like signals (Karnovsky and Klymkowsky, 1995; Sokol et al., 1995; Molenaar et al., 1996; Sokol, 1996; Yang-Snyder et al., 1996; Yost et al., 1996; Zeng et al., 1997; Behrens et al., 1998) .
Wg plays a crucially important role in the development of the Drosophila imaginal discs (Baker, 1988; Simcox et al., 1989; Couso et al., 1993) . The wing disc in particular requires Wg at several phases of its development. In the absence of Wg activity, both the wing blade and the hinge are deleted and replaced by notum (Sharma and Chopra, 1976; Morata and Lawrence, 1977) . The early expression of wg during the second larval instar is required to establish the wing primordium (Couso et al., 1993; Williams et al., 1993; Ng et al., 1996) . During the second instar, Wg collaborates with Notch to activate and maintain the expression of vestigial (vg) and scalloped (sd), which are essential for the growth of the wing (Williams et al., 1991; Williams et al., 1994; Couso et al., 1995; Kim et al., 1996; Halder et al., 1998; Simmonds et al., 1998; Klein and Arias, 1999; Varadarajan and VijayRaghavan, 1999) . During the third instar at the D/V boundary, Wg and Notch signal to generate the elaborate sensory organs that decorate the adult wing margin (Couso et al., 1994; Williams et al., 1994; Diaz-Benjumea and Cohen, 1995; Rulifson and Blair, 1995; Neumann and Cohen, 1996b) .
The role of Wg in the development of the Drosophila wing has been extensively studied, but the role of Wg in the development of the wing hinge has been less well studied. Wg is important for hinge development (Neumann and Cohen, 1996a; Klein and Arias, 1998) . However, Wg does not signal through vg in the wing hinge, and the exact role of Wg in the development of the hinge is still uncertain.
In this paper we have undertaken a study of the role of Wg in the proximal
wing hinge. We have used overexpression and ectopic expression studies and
loss-of-function experiments to determine the role of Wg in the development
of the wing hinge. We show that the dorsal hinge requires Wg for the establishment
of Axillary Sclerite 3, and the ventral hinge requires Wg to restrict the
formation of the Axillary Pouch.
Materials and Methods
Fly Strains: The following GAL4 lines were the main ones used in this study: 30A GAL4/CyO (Brand and Perrimon, 1993) , MS1096 GAL4 (Capdevila and Guerrero, 1994) , and decapentaplegic GAL4/TM6b (Staehling-Hampton et al., 1994) . All GAL4 lines were used in crosses with the following UAS lines: UASwgts (Wilder and Perrimon, 1995) , UASwg (Lawrence et al., 1995) , UASzw3 (Steitz et al., 1998) , UASdsh (Axelrod et al., 1996) , UASarmS10 and UASarmDN (Pai et al., 1997) , UASpanDN (van de Wetering et al., 1997) , UASDfz2 and UASDfz2-GPI (Cadigan et al., 1998) . The progeny of crosses between GAL4 and UAS lines is written as GAL4 - UAS. Canton-S and w1118 were used as wild-type stocks. To visualize the expression pattern of these GAL4 lines in imaginal discs from third instar larvae, each GAL4 line was crossed to UASlacZ (Brand and Perrimon, 1993) , and the discs from the larvae produced by these crosses were stained with X-Gal (see below). All crosses were done at 25 ºC unless otherwise specified. All crosses with UASwgts were kept at 25 ºC for the first day of development and then downshifted to 18 ºC for specified periods of time.
For experiments with wgts (wgIL114; Nusslein-Volhard et al., 1984) , wgts pr bw / CyO ftz-lacZ flies were crossed to wgCX4 b pr / CyO hb-lacZ (Baker, 1987; Struhl et al., 1992) , and the progeny kept at 18°C until late second/early third larval instar, and then upshifted to 25°C for 20-24 hours. wgCX4 is a null allele of wg that contains a 2.7-kb deletion that completely removes the first exon (van den Heuvel et al., 1993) .
Mosaic clones of dsh were induced using the y1 w1118 dsh75 FRT18A chromosome as described by Klingensmith et al., (1994) . Similarly, clones of zw3 were made as described by Wilder and Perrimon ( 1995) by employing the y1 sggD127 v f FRT18A chromosome. Clones of wgCX4 were induced as described by Neumann and Cohen (1997) by utilizing the wgCX4 FRT40A chromosome. Mosaic clones were identified by means of the presence of the y mutant phenotype, which causes bristles to have a lighter color.
Direct Flight Muscles were observed with myosin rod protein-lacZ
(Standiford et al., 1997)
.
Immunohistochemistry: b-galactosidase (b-Gal) staining of imaginal discs was done according to the protocol of Ashburner (1989) .
In situ hybridization of imaginal discs was done with DIG-labeled probes of wg, according to the protocol of Sturtevant and Bier (1993) .
b-Gal staining of thoraces for DFM examination was done by dissecting adult thoraces and staining with X-Gal using the Ashburner protocol. For designation of individual the Direct Flight Muscles (DFMs), we followed the nomenclature and anatomical description of Miller (1950) .
To stain imaginal discs with anti-b-Gal antibodies, we employed the
protocol of Campbell et al.,
(1993)
and used a rabbit anti-b-Gal at 1:500 (5 Prime®3 Prime, Inc.). To stain
Direct Flight Muscles with a rabbit anti-Myosin Rod Protein (MRP) antibody,
we followed the protocol of Standiford et al.
(1997)
, and also used this antibody at 1:500.
Wing Hinge
Preparations: All unstained wing hinges
were dissected in 70% ethanol and mounted in Hoyer’s Medium
(Ashburner, 1989)
between two cover slips so that both the dorsal and ventral sides could be
examined. The preparations were then baked at 60 °C and pressed with weights
to make them flat. For the wing hinge, the nomenclature of Bryant
(1978)
was used.
Results
In late third-instar wing imaginal discs, the wg gene is transcribed in a stripe of cells across the D/V boundary, and in two concentric rings that surround the wing pouch (Baker, 1988; Couso et al., 1993; Williams et al., 1993 ; Figure 1A-B). The central stripe of wg-expressing cells gives rise to the wing margin (Couso et al., 1994; Rulifson and Blair, 1995) . The inner ring of wg gives rise to distal hinge structures like the Costa, Dorsal Radius and Alula (Neumann and Cohen, 1996a) . The outer ring of wg expression maps to the proximal hinge, which generates the Axillary Sclerites, Axillary Chord, and various structures of the ventral hinge (Figure 1B; see below). More proximally lies the primordium of the mesonotum, and a stripe of wg expression in this region, elaborated during the third larval instar, helps to establish the pattern of sensory bristles (Phillips and Whittle, 1993) .
To ascertain the effects of Wg in the proximal wing hinge, we used several GAL4 lines to drive expression in the hinge. GAL4 line 30A (Brand and Perrimon, 1993) drives expression throughout the dorsal and ventral hinge primordia, but not in the wing pouch (Figure 1C). dppGAL4 (Staehling-Hampton et al., 1994) drives expression along the anterior/posterior border, including the medial portion of the dorsal hinge, and to a lesser extent the ventral hinge (Figure 1D). MS1096 (Capdevila and Guerrero, 1994) drives expression in the dorsal hinge and the dorsal and ventral wing pouch but is excluded from the ventral hinge (Figure 1E). Similar effects were observed with different GAL4 lines.
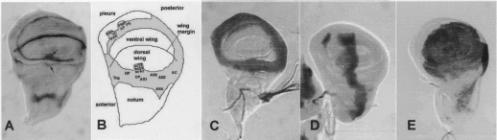 |
| Figure 1. (A) The expression patterns of wg in the wing imaginal disc from late third-instar larvae, as ascertained by in situ hybridization. wg is expressed in a stripe that corresponds to the future wing margin, and in the hinge primordium (see arrows) in two concentric rings that encircle the wing pouch (Baker, 1988; Couso et al., 1993; Phillips and Whittle, 1993; Williams et al., 1993; Neumann and Cohen, 1996a) . wg is also expressed in a stripe in the presumptive notum (Phillips and Whittle, 1993) . (B) Fate map of the Drosophila wing imaginal disc from a third instar larvae (modified from Bryant, 1975). The adult wing blade, notum, and wing hinge of Drosophila all develop from the wing imaginal disc. The gray region depicts the hinge anlage and the notal, wing and pleural anlagen are all in white. The abbreviations used in this fate map are as follows: AC - Axillary Chord; AP - Axillary Pouch; AS1 - Axillary Sclerite 1; AS2 - Axillary Sclerite 2; AS3 - Axillary Sclerite 3; AS4 - Axillary Sclerite 4; HP - Humeral Plate; PAA - Postalar Apophysis; PS - Pleural Sclerite; PVR - Proximal Ventral Radius; PWP - Pleural Wing Process; sc12 - sensilla campaniformia 12, sc25; sensilla campaniformia 25; sc4d - sensilla campaniformia 4 on the dorsal side; Teg - Tegula; UP - Unnamed Plate; YC - Yellow Club. (C) Wing imaginal disc from a 30A - UASlacZ larva, stained with X-GAL. This GAL4 insertion drives expression throughout the dorsal and ventral hinge anlagen. (D) Wing disc from a dppGAL4 - UASlacZ larva stained with X-GAL. This line drives expression along the anterior/posterior border, including the medial region of the dorsal hinge anlage, and in the ventral hinge to a lesser extent. (E) Wing disc from a MS1096 - UASlacZ larva stained with X-GAL. This line drives expression in the dorsal hinge anlage, and the dorsal and ventral wing pouch as well. It is excluded from the ventral hinge anlage. |
Excessive Wg activity causes
duplication of AS3 in the dorsal wing hinge.
Misexpression of wg in the hinge produces dramatic phenotypes, primarily characterized by duplication of AS3 with concomitant reduction or elimination of AS1, AS2, and the UP (Figure 2C-D). While in most cases it seems that duplication of AS3 occurs at the expense of the other structures, we have observed diminution or loss of AS1, AS2, and the UP without duplication of AS3, especially at lower levels of ectopic wg expression. AS4 is also sometimes deformed, apparently as a consequence of its detachment from AS3. Duplication or loss of these cuticular elements also results in duplication or loss of the direct flight muscles that attach to them (not shown).
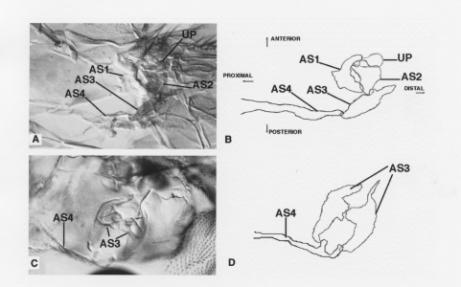 |
| Figure 2. (A) Dorsal hinge from a wild-type adult Drosophila. In all hinge photographs, anterior is up, and proximal is to the left. (B) A cartoon tracing of the salient features found in Figure 2A. The cartoon tracings sometimes represent a synthesis of multiple focal planes. (C) A dorsal hinge from a dppGAL4 - UASwg pharate adult grown at 25 °C until the early third instar, and then downshifted to 18 °C for the remainder of its larval/pupal life (n = 23). AS1, AS2, and the UP have all been removed and AS3 is duplicated. Sometimes AS1, AS2, and the UP are missing without duplication of AS3 (13%; not shown). (D) A cartoon tracing of Figure 2C. |
Loss of zw3 mimics ectopic wg activation in imaginal discs (Diaz-Benjumea and Cohen, 1994; Wilder and Perrimon, 1995) . Large mitotic clones of zw3 that cover most of the dorsal hinge cause duplication of AS3 at the expense of AS1, AS2 and the UP (not shown).
Excessive Wg activity eliminates
the Axillary Pouch in the ventral hinge.
The ventral hinge contains several cuticular landmarks that are easily recognizable (Figure 3A-B). For our analysis, the two most relevant structures are the Axillary Pouch (AP) and the Pleural Sclerite (PS). The AP is a cuticular pouch that is recognizable by its size and by the hairs that cover it. The PS is a plate that juxtaposes the AP.
Misexpression of wg causes loss of the AP and expansion of the PS (Figure 3C-D). Loss or reduction of the AP is the most reproducible effect of ectopically expressing or overexpressing wg in the ventral wing hinge. Mitotic clones of zw3 that cover this portion of the ventral hinge also cause reduction or loss of the AP, depending on the size of the clone (not shown).
Overexpression of dsh, or a constitutively activated armadillo allele (armS10) also produces phenotypes that are indistinguishable from those generated by the ectopic expression of wg (not shown).
Reduction of Wg signaling in
the dorsal wing hinge removes AS3.
A temperature-sensitive allele of wg (wgIL114) was employed in order to assay the effect of lowering Wg activity in the hinge. If wgIL114 / wgCX4 animals were upshifted to the restrictive temperature during the late second or early third larval instar, such animals produced pharate adults with hinge defects that consistently showed reductions of AS3 (Figure 4 A-B).
dsh mutant clones induced during the first instar exhibit dramatic phenotypes in the dorsal hinge. Dorsal hinge structures, with the exception of AS1, are obliterated if they fall within the clone (not shown). Mitotic clones of wgCX4 do not cause hinge phenotypes (not shown), since the surrounding wild-type tissue secretes Wg and non-autonomously rescues the mutant tissue (Li et al., 1995) .
Phenotypes produced by overexpression of zw3
(Steitz et al., 1998)
or panDN
(van de Wetering et al., 1997)
are generally opposite in nature to those produced by misexpression of wg. Overexpression of zw3 causes reduction or elimination of AS3 (Figure 4C-D),
as does ectopic expression of Dfz2-GPI or panDN (not shown).
Reduction of Wg signaling in
the ventral hinge causes duplication of the AP.
Ventrally, since hyperactivation of Wg signaling in the ventral hinge causes loss of the AP, reduction or elimination of Wg signaling in the ventral hinge should cause enlargement or duplication of the AP. Mitotic clones of dsh that are large enough to include the proximal hinge result in duplication of the AP and the YC (Figure 4E-F). Phenotypes resulting from expression in more restricted domains were consistent with cell autonomous effects of this gene (not shown).
Ectopic expression of zw3, which
attenuates Wg activity, also causes duplication of the AP (Figure 4G-H). Ectopic
expression of Dfz2-GPI or panDN produced
similar results (not shown). These results are consistent with the mediation
of Wg signaling by Dsh, Arm, Pan, and Zw3 in the hinge.
Discussion
Analysis of the phenotypes induced by hyperactivation or attenuation of Wg activity in the wing hinge has shown that Wg is required for the formation of AS3 in the dorsal hinge and the restriction of the AP in the ventral hinge.
In the wing hinge, the Wg protein seems to have three distinct tasks: establishment of the wing field, delineation of the hinge anlage from the wing anlage, and establishment of specific structures in the proximal and distal portions of the hinge.
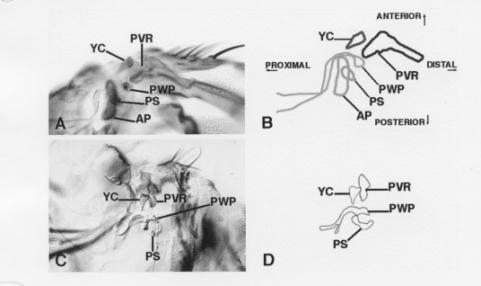 |
| Figure 3. (A) Ventral hinge from a wild-type adult. Ventrally, the confluence of wing veins #1, #2, and #3 terminates in a structure called the Proximal Ventral Radius (PVR), which bears three groups of sensory structures that contain four (sc4v), three (sc3) and five (sc5) campaniform sensillae respectively (Bryant, 1978) . Just proximal to the PVR is the Yellow Club (YC), which is the most conspicuous ventral hinge structure. Posterior to the YC is a series of plates and a pouch. The Axillary Pouch (AP) is a large outpouching of the hinge cuticle, which is covered by hairs. Just anterior to the AP lies the Pleural Wing Process (PWP). The PWP arches around the AP to form the pleural suture, which separates the pleura into the mesopleura and the pteropleura. The head of the PWP articulates with the ventral side of AS2. Posterior to the PWP is the Pleural Sclerite (PS). The PS is hidden from view in some preparations by the AP, which overlies and obscures it. (B) A cartoon tracing of Figure 3A. (C) Ventral hinge from a 30A - UASwg pharate adult. The AP is missing and the PS is enlarged. (D) A cartoon tracing of Figure 3C. |
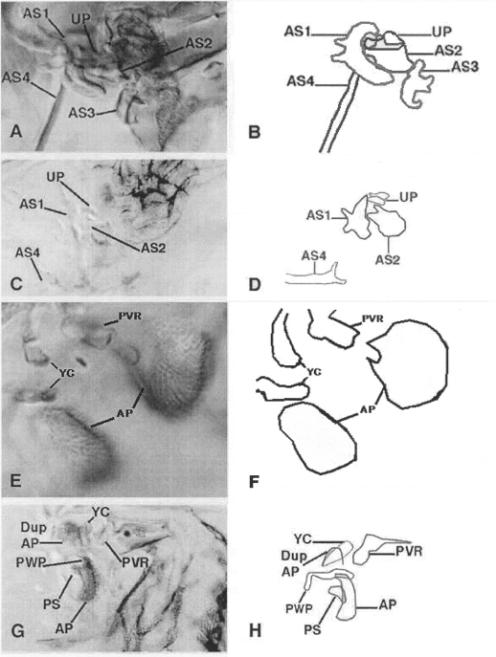 |
| Figure 4. (A) A dorsal hinge from a wgts / wgCX4 pharate adult that was grown as described (See Materials and Methods). All proximal hinge elements are normal with the exception of AS3, which is smaller than normal and fails to articulate with AS4 (n = 13). (B) A cartoon tracing of Figure 4A. (C) Dorsal wing hinge from a MS1096 - UASzw3 adult (n = 31). AS3 is missing, and AS1, AS2, AS4, and the UP are unaffected. Wings from MS1096 - UASzw3 animals are very small and devoid of marginal structures (data not shown). (D) A cartoon tracing of Figure 4C. (E) Ventral wing hinge from a y1 w1118 dsh75 FRT18A / w1118 arm-lacZ FRT18A; hsflp38 / + animal, in which a large dsh clone has been induced. The AP and YC are both duplicated and the PWP and PS are missing (n = 12). (F) A cartoon tracing of Figure 4E. (G) A ventral hinge from a dppGAL4 - UASzw3 pharate adult (n = 21). All the ventral hinge structures are intact, but the AP is duplicated. The duplicate AP lies just posterior to the PAA. (H) A cartoon tracing of Figure 4G. |
Wg is required for the establishment
of the wing field.
During the second instar, wg
is expressed in a patch of cells nested within the anterior and ventral portions
of the wing disc
(Couso et al., 1993; Williams et
al., 1993; Ng et al., 1996)
. Without Wg activity during the second instar, the entire wing field, both
the wing blade and the hinge, is absent and is replaced by notum
(Sharma and Chopra, 1976; Morata
and Lawrence, 1977)
. Therefore Wg is essential to establish the anlagen that give rise to both
the wing pouch and the hinge primordium.
Wg is necessary for delineation
between the wing and hinge primordia.
Wg helps establish the wing blade by directing the expression of the wing-specific gene vg. Proper activation of vg in the wing pouch requires a collaboration between Wg, the Notch receptor, which is activated by interactions between dorsal and ventral cells at the D/V boundary (Williams et al., 1994; Kim et al., 1995; Kim et al., 1996) , and Decapentaplegic (Dpp), the TGF-β ortholog in Drosophila (Padgett et al., 1987) , which is secreted by the cells along the anterior/posterior (A/P) border (Blair, 1994; Kim et al., 1996; Zecca et al., 1996; Kim et al., 1997; Neumann and Cohen, 1997) . Wg, which is secreted from the D/V boundary cells, induces vg expression at a distance from the D/V boundary (Zecca et al., 1996; Neumann and Cohen, 1997) . This generates a gradient of Vg, with peak levels at the D/V boundary.
Outside the wing pouch, Wg activates targets other than vg. One of the targets of Wg signaling within the hinge is the hth gene, which, during the third instar, shows an expression pattern that is restricted to the hinge primordia and is required for hinge development (Azpiazu and Morata, 2000; Casares and Mann, 2000) . Clones of hth and extradenticle (exd), show similar effects in the hinge (Gonzalez-Crespo and Morata, 1995; Rauskolb et al., 1995) . In imaginal discs, extradenticle shows uniform expression (Rauskolb et al., 1995) but is only active in those cells where it shows nuclear localization (Mann and Abu-Shaar, 1996; Aspland and White, 1997) , and Hth activates the nuclear localization of Exd (Rieckhof et al., 1997; Pai et al., 1998) . Presently, it is not clear if Hth has any function independent of Exd.
Wg and Dpp activate the expression of the vg gene, and in the wing pouch, Vg represses hth expression (Azpiazu and Morata, 2000) . Initially, the wing disc, which is derived from the second thoracic segment, shows high and uniform levels of hth expression (Rieckhof et al., 1997) . The repression of hth by Vg prevents the nuclear accumulation of Exd in the cells of the wing pouch, which prevents the wing pouch cells from forming more proximal structures. Therefore, Wg establishes the hinge by activating hth in the hinge and directing the expression of Vg, which prevents the expression of hth, in the wing. In this way, Wg helps delineate between the hinge and wing primordia.
The specification of notum rather than hinge appears to come from the
notum-specific expression of the Iroquois-C
(Iro-C) gene
(Diez del Corral et al., 1999)
.
Wg establishes specific structures
in the proximal and distal hinge.
Once the wing field has been established and the hinge primordium delineated, Wg, Hth and another gene that shows hinge-specific expression in the wing disc called teashirt (tsh; Fasano et al., 1991; Soanes et al., 2001) distinguish the distal and proximal regions of the hinge. In the hinge, Wg and Tsh seem to maintain the high levels of hth expression that were inherited from the embryo (Azpiazu and Morata, 2000; Casares and Mann, 2000) . Expression of tsh is restricted to the proximal hinge, and it is possible that the combination of Tsh, Wg and Hth generates the proximal hinge, whereas the distal hinge is specified by the combination of Wg, Hth, and some of the wing pouch-specific genes whose expression domains spill into the distal hinge, like nubbin (Azpiazu and Morata, 2000) .
Within these two molecular contexts, Wg specifies the development of particular hinge structures. The inner ring of wg-expressing cells is missing in wing imaginal discs from animals homozygous for wgspd-fg. Flies homozygous for wgspd-fg or wgIL114 / wgCX4 animals grown at the restrictive temperature during the third instar, show deletion of part of the Dorsal Radius, Costa and Alula (Phillips and Whittle, 1993; Neumann and Cohen, 1996a) .
Likewise, if Wg activity is attenuated during development of the proximal
hinge, AS3 fails to develop and the AP duplicates. While other axillary sclerites
in the dorsal hinge are also affected by the absence of Wg signaling, AS3
is the most sensitive. The same can be said for the YC in the ventral hinge,
which can also be affected by levels of Wg signaling, but is much less sensitive
than the AP (data not shown). Therefore, even though AS3 and the AP are the
structures most readily affected by variations in Wg signaling, they are by
no means the only structures that respond to Wg, and the role of Wg in the
proximal hinge is almost certainly more complex than just the specification
of these two structures.
Wg does not function simply
as a mitogen in the developing hinge.
Neumann and Cohen (1996a) concluded that Wg acts as a mitogen for the hinge since overexpression of Wg causes enlargement of the hinge, and a decrease in Wg expression in the hinge causes loss of hinge tissue. Our results are essentially consistent with their conclusions, but we find that the mitogenic effect of Wg only applies to specific areas of the hinge, since other regions of the hinge do not overproliferate in response to ectopic Wg. AS1, AS2, the AP, and the UP shrink or disappear rather than enlarge when Wg is ectopically expressed. In addition, duplication of AS3, which is a characteristic result of ectopically expressing Wg with dppGAL4, occurs at the expense of these other structures in the dorsal hinge. Ventrally, several structures appear to require Wg activity in order to form, but, with the exception of the PS, overexpression of Wg does not enlarge them. Dorsally, the primary role of Wg seems to be in the formation of AS3 and sc12 (Neumann and Cohen, 1996a).
Our data indicate that even in cells where Wg apparently increases
proliferation, it is also associated with changes in cell fate. Therefore,
the role of Wg in the development of the hinge is complex, and includes mitogenic
and cell determination activities.
Concluding Remarks
The wing hinge of Drosophila is a tissue that requires Wg for its development. Particularly two of its structures; AS3 in the dorsal hinge requires Wg for its development, and the AP in the ventral hinge requires Wg for restriction of its development. It is particularly interesting that portions of the hinge respond differently to this protein, but the molecular contexts in which Wg is expressed probably determine the effects it has on the cells that experience it.
Acknowledgments: We thank Robert Riddle at U of Penn for his comments and input while this work was in progress. We are especially indebted to Steve DiNardo and David Standiford at U of Penn and Charles Morrisey at SAU for their helpful comments on the manuscript. This work was supported by the University of Pennsylvania, the University of Pennsylvania Research Foundation and by the National Institutes of Health under Grant No. GM55162.
References:
Ashburner, M., 1989, Drosophila: A Laboratory Manual.
Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press; Aspland, S. E. and White, R. A., 1997,
Development 124: 741-7; Axelrod,
J. D., Matsuno, K., Artavanis-Tsakonas, S. and Perrimon, N., 1996, Science
271: 1826-32; Azpiazu, N. and
Morata, G., 2000, Development 127: 2685-93;
Baker, N. E., 1987, EMBO J. 6: 1765-73; Baker, N. E., 1988, Development 102: 489-97; Behrens, J., Jerchow, B. A., Würtele,
M., Grimm, J., Asbrand, C., Wirtz, R., Kühl, M., Wedlich, D. and Birchmeier,
W., 1998, Science 280: 596-99; Bhanot, P., Brink, M., Samos, C. H., Hsieh, J. C., Wang, Y.,
Macke, J. P., Andrew, D., Nathans, J. and Nusse, R., 1996, Nature 382: 225-30;
Blair, S. S., 1994, Dev Biol 162: 229-44;
Brand, A. and Perrimon, N., 1993, Development 118: 401-15; Brunner, E., Peter, O., Schweizer, L. and Basler, K., 1997,
Nature 385: 829-33; Bryant, P.
J., 1978, Pattern Formation in Imaginal Discs. In The Genetics and Biology
of Drosophila, vol. 2C ed. M. Ashburner and T. R. F. Wright), pp. 229-335.
New York: Academic Press; Cadigan,
K. M., Fish, M. P., Rulifson, E. J. and Nusse, R., 1998, Cell 93: 767-77; Cadigan, K. M. and Nusse, R., 1997, Genes
and Development 11: 3286-305; Campbell,
G., Weaver, T. and Tomlinson, A., 1993, Cell 74: 1113-23; Capdevila, J. and Guerrero, I., 1994,
EMBO 13: 4459-68; Casares, F.
and Mann, R. S., 2000, Development 127: 1499-508; Couso, J. P., Bate, M. and Martinez, A. A., 1993, Science 259,
484-89; Couso, J. P., Bishop,
S. A. and Martinez Arias, A., 1994, Development 120: 621-36; Couso, J. P., Knust, E. and Martinez Arias,
A., 1995, Curr Biol 5: 1437-48; Diaz-Benjumea,
F. J. and Cohen, S. M., 1994, Development 120: 1661-70;
Diaz-Benjumea, F. J. and Cohen, S. M., 1995, Development 121: 4215-25; Diez del Corral, R., Aroca, P., JL, G. m.-S., Cavodeassi, F.
and Modolell, J., 1999, Genes Dev 13: 1754-61; Fasano, L., Roder, L., Core, N., Alexandre, E., Vola, C., Jacq,
B. and Kerridge, S., 1991, Cell 64: 63-79; Gonzalez-Crespo, S. and Morata, G., 1995, Development 121:
2117-25; Halder, G., Polaczyk,
P., Kraus, M. E., Hudson, A., Kim, J., Laughon, A. and Carroll, S., 1998,
Genes Dev 12: 3900-9; Hamada,
F., Tomoyasu, Y., Takatsu, Y., Nakamura, M., Nagai, S., Suzuki, A., Fujita,
F., Shibuya, H., Toyoshima, K., Ueno, N. et al.,
1999, Science 283: 1739-42; Karnovsky,
A. and Klymkowsky, M. W., 1995, Proc. Natl. Acad. Sci. USA 92: 4522-6;
Kim, J., Irvine, K. D. and Carroll, S. B., 1995, Cell 82: 795-802; Kim, J., Johnson, K., Chen, H. J., Carroll, S. and Laughon,
A., 1997, Nature 388: 304-8; Kim,
J., Sebring, A., Esch, J. J., Kraus, M. E., Vorwerk, K., Magee, J. and Carroll,
S. B., 1996, Nature 382: 133-8; Klein,
T. and Arias, A. M., 1998, Dev Biol 194: 196-212; Klein, T. and Arias, A. M., 1999, Development
126: 913-25; Klingensmith, J.,
Nusse, R. and Perrimon, N., 1994, Genes and Development 8: 118-30;
Lawrence, P. A., Bodmer, R. and Vincent, J. P., 1995, Development 121:
4303-08; Li, W., Ohlmeyer, J. T., Lane, M. E. and
Kalderon, D., 1995, Cell 80: 553-62;
Mann, R. S. and Abu-Shaar, M., 1996, Nature 383: 630-3; Miller, A., 1950, The internal anatomy
and histology of the imago of Drosophila melanogaster. In: Biology of Drosophila, (Demerec,. M.,
ed.), pp. 424-42. New York: Wiley; Miyan,
J. A. and Ewing, A. W., 1985, Philosophical Transactions of the Royal Society,
London 311B: 271-302; Molenaar,
M., van de Wetering, M., Oosterwegel, M., Peterson-Maduro, J., Godsave, S.,
Korinek, V., Roose, J., Destree, O. and Clevers, H., 1996, Cell 86: 391-99;
Morata, G. and Lawrence, P. A., 1977, Dev Biol 56: 227-40; Neumann, C. J. and Cohen, S. M., 1996a, Development 122: 1781-9;
Neumann, C. J. and Cohen, S. M., 1996b, Development 122: 3477-85;
Neumann, C. J. and Cohen, S. M., 1997, Development 124: 871-80; Ng, M., Diaz-Benjumea, F. J., Vincent,
J. P., Wu, J. and Cohen, S. M., 1996, Nature 381: 316-8; Nusslein-Volhard, C., Wieschaus, E. and
Kluding, H., 1984, Roux's Archives of Developmental Biology 193: 267-82; Padgett, R. W., St Johnston, R. D. and
Gelbart, W. M., 1987, Nature 325: 81-4;
Pai, C.-Y., Kuo, T., Jaw, T., Kurant, E., Chen, C., Bessarah, D., Salzberg,
A. and Sun, Y., 1998, Genes and Development 12: 435-46; Pai, L. M., Orsulic, S., Bejsovec, A.
and Peifer, M., 1997, Development 124: 2255-66; Peifer, M., Pai, L. M. and Casey, M., 1994a, Dev Biol 166:
543-56; Peifer, M., Rauskolb,
C., Williams, M., Riggleman, B. and Wieschaus, E., 1991, Development 111:
1029-43; Peifer, M., Sweeton,
D., Casey, M. and Wieschaus, E., 1994b, Development 120: 369-80; Peifer, M. and Wieschaus, E., 1990, Cell
63: 1167-76; Phillips, R. G.
and Whittle, J. R., 1993, Development 118: 427-38; Rauskolb, C., Smith, K., Peifer, M. and Wieschaus, E., 1995,
Development 121: 3663-71; Rieckhof,
G., Casares, F., Ryoo, H. D., Abu-Shaar, M. and Mann, R. S., 1997, Cell 91:
171-83; Ruel, L., Stambolic,
V., Ali, A., Manoukian, A. S. and Woodgett, J. R., 1999, J Biol Chem 274:
21790-6; Rulifson, E. J. and
Blair, S. S., 1995, Development 121: 2813-24; Sharma, R. P. and Chopra, V. L., 1976, Dev Biol 48: 461-5;
Siegfried, E., Chou, T. B. and Perrimon, N., 1992, Cell 71: 1167-79;
Siegfried, E., Perkins, L. A., Capaci, T. M. and Perrimon, N., 1990,
Nature 345: 825-29; Simcox, A.
A., Roberts, I. J. H., Hersperger, E., Gribbin, M. C. and Whittle, J. R. S.,
1989, Development 107: 715-22; Simmonds,
A. J., Liu, X., Soanes, K. H., Krause, H. M., Irvine, K. D. and Bell, J. B.,
1998, Genes Dev 12: 3815-20; Soanes,
K. H., MacKay, J. O., Core, N., Heslip, T., Kerridge, S. and Bell, J. B.,
2001, Mech Dev 105: 145-51; Sokol,
S. Y., 1996, Current Biology 6: 1456-67; Sokol, S. Y., Klingensmith, J., Perrimon,
N. and Itoh, K., 1995, Development 121: 1637-47; Staehling-Hampton, K., Jackson, P. D.,
Clark, M. J., Brand, A. H. and Hoffmann, F. M., 1994, Cell Growth Diff. 5:
585-93; Standiford, D. M., Davis,
M. B., Miedema, K., Franzini-Armstrong, C. and Emerson, C. P., Jr., 1997,
Journal of Molecular Biology 265: 40-55;
Steitz, M. C., Wickenheisser, J. K. and Siegfried, E., 1998, Developmental
Biology 197, 218-33; Struhl,
G., Johnston, P. and Lawrence, P. A., 1992, Cell 69: 237-49;
Sturtevant, M. A., Roark, M. and Bier, E., 1993, Genes and Development
7: 961-73; Theisen, H., Purcell, J., Bennett, M.,
Kansagara, D., Syed, A. and Marsh, J. L., 1994, Development 120: 347-60; Tolwinski, N. S. and Wieschaus, E., 2001,
Development 128: 2107-17; van
de Wetering, M., Cavallo, R., Dooijes, D., van Beest, M., van Es, J., Loureiro,
J., Ypma, A., Hursh, D., Jones, T., Bejsovec, A. et al.,
1997, Cell 88: 789-99; van den Heuvel, M., Harryman-Samos, C.,
Klingensmith, J., Perrimon, N. and Nusse, R., 1993, Embo J 12: 5293-302; Varadarajan, S. and VijayRaghavan, K.,
1999, Dev Genes Evol 209: 10-7; Wilder,
E. L. and Perrimon, N., 1995, Development 121: 477-88; Willert, K., Logan, C. Y., Arora, A.,
Fish, M. and Nusse, R., 1999, Development 126: 4165-73; Williams, J. A., Bell, J. B. and Carroll,
S. B., 1991, Genes Dev 5: 2481-95; Williams, J. A., Paddock, S. W. and Carroll, S. B., 1993, Development
117: 571-84; Williams, J. A.,
Paddock, S. W., Vorwerk, K. and Carroll, S. B., 1994, Nature 368: 299-305;
Wisser, A., 1987, Zoomorph. 106: 261-68;
Wisser, A., 1988, Zoomorph. 107: 359-69;
Yang-Snyder, J., Miller, J. R., Brown, J. D., Lai, C. J. and Moon,
R. T., 1996, Current Biology 6: 1302-06;
Yost, C., Torres, M., Miller, J. R., Huang, E., Kimelman, D. and Moon,
R. T., 1996, Genes and Development 10: 1443-54; Zecca, M., Basler, K. and Struhl, G., 1996, Cell 87: 833-44;
Zeng, L., Fagotto, F., Zhang, T., Hsu, W., Vasicek, T. J., Perry, W.
L., 3rd, Lee, J. J., Tilghman, S. M., Gumbiner, B. M. and Costantini, F.,
1997, Cell 90: 181-92.