In phase-transfer catalysis, the reaction is carried out in a biphasic system, often water and a hydrophobic organic liquid, in which surfactants (quaternary ammonium salts) are added in order to enhance the transfer of molecules between phases, increase the interfacial surface area (emulsification), and improve selectivity[1]. This approach is of
special interest when valuable products formed upon reaction can be separated into the other phase, based on their solubility, preventing undesired secondary reactions (e.g. thermal decomposition). More broadly, ongoing partitioning of by-products on the basis of their relative solubility can result in substantial simplifications at the isolation and purification stages, obviating the need for procedures such as distillation that might damage heat-sensitive compounds.
However, a major drawback of this technique is the separation of the surfactant molecules from the reaction mixture, which requires expensive separation methods, such as membrane filtration[2]. This disadvantage can be overcome by replacing the surfactants with amphiphilic catalyst particles that can be readily recovered and reused after each
catalytic run. This novel methodology combines the advantages of phase transfer and heterogeneous catalysis: a) increased interfacial area, b) enhanced mass transfer of molecules between the two phases, c) simplified reaction/separation process by using a recoverable solid catalyst instead of surfactant, and d) effective separation of products from the reaction mixture by differences in the water-oil solubility (avoiding heating that leads to product decomposition).
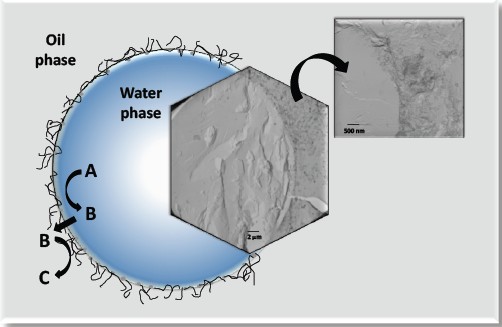 |
| Figure 1. TEM images of the interface in a water-in-oil emulsion droplet stabilized by SWCNT–SiO2,together with a general reaction scheme. |
In this context, we have recently shown that carbon nanotubes (hydrophobic) fused to oxide nanoparticles (hydrophilic) are able to stabilize Pickering emulsions and, when properly functionalized, catalyze reactions at the water/oil interface (i.e., liquid/solid/liquid interfacial catalysis)[3-5]. The amphiphilic character of these nanohybrid materials naturally segregates them to the water/oil interface in a biphasic system, and makes them suitable for the stabilization of emulsions with small droplet sizes with remarkable stability.3-5 We have also demonstrated that the hydrophilic/lipophilic balance (HLB) can be modified by tailoring the composition and structure of the CNT-metal
oxide nanoparticle. This fine control of the HLB allowed us to systematically change the type of emulsion, the droplet size, and the volume fraction of the emulsion in the water/oil mixture[3,4].
Additionally, by anchoring metal particles on the surface of these nanohybrids, as well as by using nanohybrids composed of CNT fused to basic metal oxides (e.g. MgO), metal-catalyzed as well as base-catalyzed reactions have been performed at the water/oil interface in emulsion systems[4]. The reactions with this novel methodology could have a major impact in the field of biomass conversion to fuels[6] (upgrading of pyrolysis oil and sugars), production of specialty chemicals and pharmaceuticals and Fischer-Tropsch.
References:
[1] C.M. Starks. J. Am. Chem. Soc. (93), 1971, 195.
[2] T.M. Schmitt. Analysis of surfactants. 2nd Ed. New York. Ed. Marcel Dekker, Inc.
2001.
[3] M. Shen, D.E. Resasco. Langmuir (25), 2009, 10843.
[4] S. Crossley, J. Faria, M. Shen, D. E. Resasco. Science (327), 2010, 68.
[5] M.P. Ruiz, J. Faria, M. Shen, S. Drexler, T. Prasomsri, D.E. Resasco. ChemSusChem,
2010, In press (DOI: 10.1002/cssc.201000322).
[6] G.W. Huber, S. Iborra, A. Corma. Chem. Rev. (106), 2006, 4044.